Cosmetic Product Safety Report (CPSR)
Introduction to Cosmetic Product Safety Report (CPSR) Understanding the requirements for selling cosmetic products in Europe and the UK is crucial for complianc... Read more.

Cosmetics check
A “cosmetics check” is essential for ensuring your cosmetic products are safe, compliant, and market-ready. At Biorius, we guide you through this pr... Read more.

EU Cosmetic Compliance – Complete Guide
EU cosmetic compliance Embarking on the journey of achieving EU cosmetic compliance can seem like navigating through uncharted waters. However, with the right g... Read more.
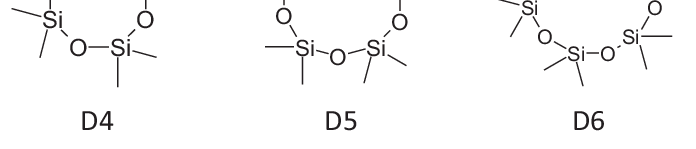
D4, D5, D6 REACh restrictions published
BIORIUS Information Letter 129: D4, D5, D6 REACh restrictions published The Commission Regulation (EU) 2024/1328 of 16 May 2024 on Cyclotetrasiloxane (D4), Cyc... Read more.

Understanding the Role of the Responsible Person (RP) in Cosmetic Regulations
Navigating the complexities of cosmetic regulations in the European Union can be a daunting task for many cosmetic brands. Central to this regulatory landscape ... Read more.

MoCRA labeling requirements
Understanding MoCRA labeling requirements MoCRA Label Requirements: The Modernization of Cosmetics Regulation Act (MoCRA) 2022 introduces significant changes to... Read more.

US Agent FDA cosmetics
Key Insights for Cosmetic Regulatory Experts US Agent FDA cosmetics: Navigating the highly regulated US cosmetic industry requires a solid understanding of FDA ... Read more.

FDA Registration for Cosmetics: A Comprehensive Guide
Understanding FDA Registration for Cosmetics under MoCRA With the enactment of the Modernization of Cosmetics Regulation Act (MoCRA), the landscape of cosmetic ... Read more.

Cosmetic Formulation: Guide for Regulatory Experts
What is a Cosmetic Formulation? A cosmetic formulation is a carefully crafted mixture of ingredients designed for application to the human body for beautifying,... Read more.

EU Fluorinated Greenhouse Gases Regulation (EU)
BIORIUS Information Letter 128 (this information letter is a follow-up to our information letter 124): Ban of fluorinated greenhouse gases in cosmetics, Regulat... Read more.

