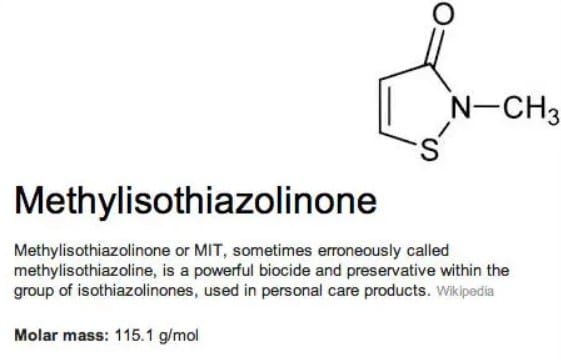
Update: Methylisothiazolinone in cosmetic products will be restricted at 15 ppm in Rinse-Off Products
July 7th, 2017
Methylisothiazolinone in cosmetic products: on July 6 th, 2017, the EU Commission published a new piece of legislation (Regulation EU No 2017/1224), which aims to restrict the use of MIT in rinse-off cosmetic products at a maximum concentration of 15 ppm (namely 0.0015 %). At this level, Methylisothiazolinone does not have a real antimicrobial activity and this ingredient should not be used to preserve finished cosmetic products any longer. However, this level allows cosmetic producers to continue the use of raw materials preserved with Methylisothiazolinone.
Contrary to the initial Regulatory Proposal, the new Regulation does not require the labeling of the specific warning ‘Contains Methylisothiazolinone’.
This new amendment to the EU Cosmetics Regulation (EC No 1223/2009) will enter into force on July 27 th, 2017 and the transitional period has been validated as follows:
- From January 27th , 2018 (6 months from now) only cosmetic products which comply with this Regulation shall be placed on the Union market.
- From April 27th , 2018 (9 months from now) only cosmetic products which comply with this Regulation shall be made available on the Union market.
Any questions?
Update: A New Regulation on Methylisothiazolinone (MIT)
July 26th, 2016
On July 22nd , 2016, the EU Commission published a new piece of legislation (Regulation EU No 2016/1198), which aims to prohibit the use of MIT in leave-on cosmetic products. This new amendment to the EU Cosmetics Regulation (EC No 1223/2009) will enter into force on August 12th, 2016 and foresees a 6-month transitional period for withdrawing non-compliant products from the Union market. In other words, only cosmetic products which comply with this Regulation shall be placed and made available on the Union market from February 12th , 2017.
The use of this preservative will remain authorized in rinse-off products at 100 ppm. However, another regulatory development aiming to limit the use of MIT at 15 ppm in rinse-off products is ongoing and should get published by May/June 2017.
For the purpose of this regulation, wet wipes are considered as leave-on products and the ban will also apply to this product category. Please, note that the use of MIT as a preservative of raw materials would also be prohibited, even at very low concentration. MIT is a ubiquitous cosmetic ingredient and many products will have to be reformulated in the next few months.
Follow-up: Methylisothiazolinone (in Rinse-Off Products) Currently under Public Consultation
April 18th , 2016
With no surprise, the EU Commission proposes to limit the use of this substance in rinse-off products at 0.0015 % (15 ppm) and to oblige the labelling of a new warning ‘Contains methylisothiazolinone’. Furthermore, they suggest a transition period of 6 months for placing compliant products on the market and 9 months for withdrawing non-compliant products from the market.
Update on Methylisothiazolinone
March 15th , 2016
Methylisothiazolinone is preservative widely used by the Cosmetic Industry, it is therefore important that you can anticipate changes related to its regulation and, when needed, adapt on time the formula of your cosmetic products.
The mixture Methylchloroisothiazolinone / Methylisothiazolinone
First, it is important to remind that the transition period for the ban of the mixture Methylchloroisothiazolinone (MCI) / Methylisothiazolinone (MIT) in leave-on cosmetic products will come to its end on April 16th, 2016.
At this date, only cosmetic products complying with the regulation EU No 1003/2014 shall be made available on the Union Market. Biorius continuously informed you about this regulatory change to come and we hope that all your products still on the EU Market have been properly reformulated. It is also useful to remind that the use of this mixture in rinse-off products is not impacted.
Methylisothiazolinone alone in leave-on cosmetic products
Then, you probably now that the Standing Committee on Cosmetic Products of the EU Commission voted the ban of MIT alone, when used in leave-on cosmetic products (including hair leave-on products). The substance being extensively used in the industry, and currently restricted at 100 ppm (0.01%) in all cosmetic products (via entry 57 of Annex V), we could have thought that this ban would come with comfortable transition periods to allow the industry to make the necessary adjustments to product formulations but this does not seem to be the case. This amendment expected from one day to the other will get enforced 20 days after its publication and then a sixmonth transition period will start. At the end of this period, only cosmetic products which comply with this Regulation shall be placed and made available on the Union market.
Methylisothiazolinone alone in rinse-off cosmetic products
Finally, the SCCS released a last Scientific Opinion on MIT recommending that this ingredient be further restricted in rinse-off products from the current 100 ppm to only 15 ppm (0.015%). The EU Commission took these recommendations on board and released a regulatory proposal reflecting the SCCS Opinion. In addition to this severe restriction, the warning “Contains Methylisothiazolinone” will probably have to be labelled. Although MIT is not effective at such a low concentration, the industry made a point of preserving a limited use of this ingredient since many raw materials are preserved with MIT (which is therefore frequently present below 10 ppm in the finished product). This regulatory change is expected to be submitted for vote at the Standing Committee in early 2017. Once voted, we can reasonably expect 3 to 6 months for the EU Commission to prepare and publish the regulation. Twenty days after the publication of this piece of legislation will start the transition period, which should be 6 months for placing on the market and 9 months for withdrawing from the market.
The recommendation of Biorius
Our recommendation is to start immediately the reformulation of your leave-on products (if not done already) and check that your raw materials suppliers are aware of this upcoming changes and ready to provide you with MIT-free alternatives. Once your formulae are EU compliant, we recommend that you revise your product label and remove the INCI name ‘Methylisothiazolinone’ from the ingredients lists of your leave-on products. The number of available preservatives is decreasing steadily and it becomes more and more difficult to find suitable alternatives. Methylparaben and Ethylparaben remain two very effective preservatives although they are not very popular those last days. Phenoxyethanol is also very good but maybe not a very safe alternative for the future (the SCCS is currently re-evaluating this ingredient). Then it exists less common and somehow less workable preservatives such as Sodium Benzoate, Potassium Sorbate, Chlorphenesin, etc. Just try to avoid Formaldehyde-releasers as their use is likely to be further restricted in the next years. Alternative solutions are typically airless packaging, single-use packaging and other technologies designed to avoid the contamination.