Comparison between the US and EU cosmetics regulations (updated in March 2026)
Cosmetics regulation in the European Union and the United States still follows two very different models in March 2026. The EU remains a highly structured pre market compliance system built around Regulation (EC) No 1223/2009, a Responsible Person (RP), a Product Information File (PIF), a Cosmetic Product Safety Report (CPSR), and CPNP (Cosmetic Products Notification Portal) notification. The United States remains less prescriptive overall, but the Modernization of Cosmetics Regulation Act of 2022 has materially strengthened FDA oversight through mandatory Facility Registration, Cosmetic Product Listing, serious Adverse Event Reporting, safety substantiation, and additional FDA authorities.
Editor’s note, March 2026: this article has been updated to reflect the current implementation status of MoCRA in the United States, including FDA Cosmetic Facility Registration, Cosmetic Product Listing, biennial facility renewal, and serious Adverse Event Reporting obligations.
The fundamental difference between the EU and U.S. cosmetics frameworks
- EU: The main difference is structural. The EU cosmetics regime is built around formal pre market compliance obligations. Before a cosmetic product is placed on the EU market, it must be supported by a safety assessment, included in a Product Information File (PIF), linked to an EU Responsible Person (RP), and notified through the CPNP (Cosmetic Products Notification Portal).
- US: The United States follows a different model. The federal framework is still less prescriptive than the EU system, but it is no longer accurate to describe it as light touch. Under MoCRA, the U.S. system now includes mandatory Cosmetic Facility Registration, Cosmetic Product Listing, serious Adverse Event Reporting, safety substantiation obligations, and broader FDA powers in specific cases. The U.S. approach remains different from the EU model, but the gap has narrowed compared with the pre MoCRA period.
European Cosmetics Regulation: Structured and Comprehensive
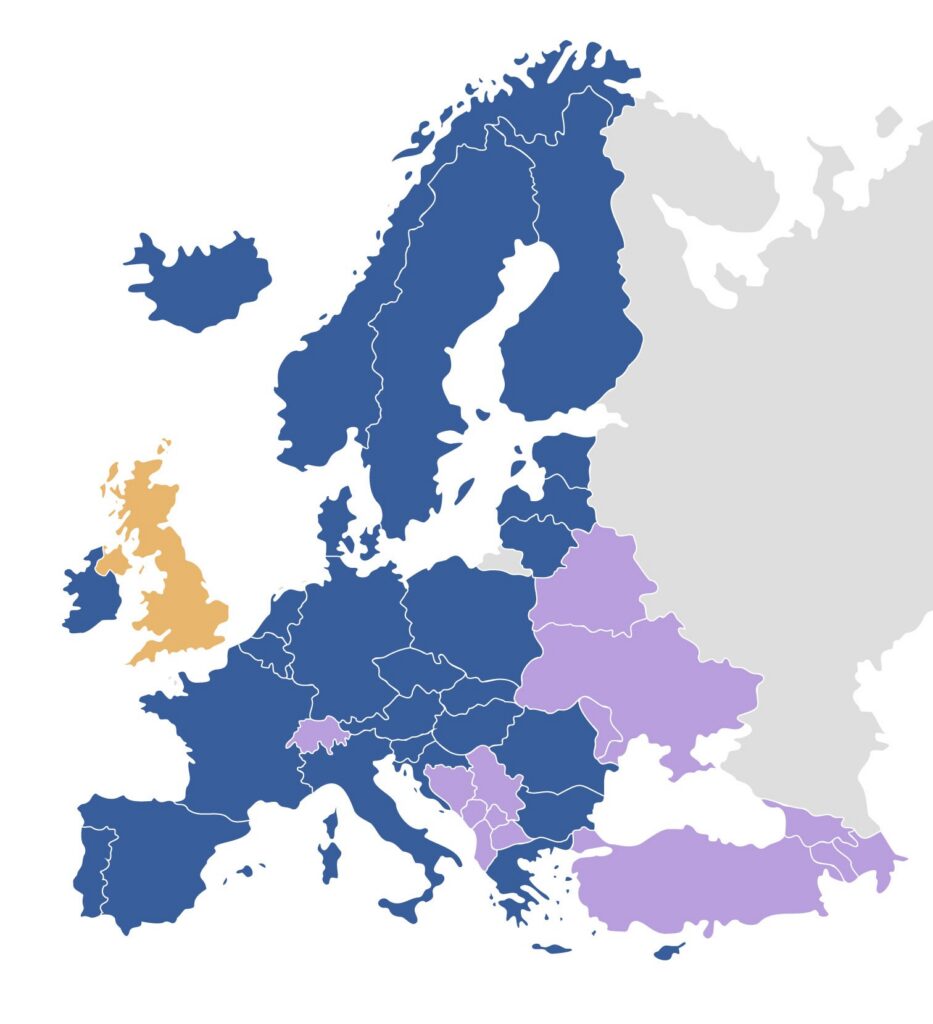
Dark blue countries: full application of the EU Cosmetics Regulation
In these countries, Regulation (EC) No 1223/2009 applies fully and directly, as they are part of the European Union single market:
Austria, Belgium, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Latvia, Liechtenstein, Lithuania, Luxembourg, Malta, Netherlands, Norway, Poland, Portugal, Romania, Slovakia, Slovenia, Spain, Sweden.
Iceland, Liechtenstein and Norway are part of the European Economic Area (EEA) and implement the EU Cosmetics Regulation with high alignment, although not as EU Member States.
Purple countries: alignment or partial alignment with the EU framework
In these countries, the EU Cosmetics Regulation does not apply directly, but national frameworks are largely aligned or inspired by it. Specific requirements, deviations, or additional local obligations may apply.
Albania, Armenia, Azerbaijan, Belarus, Bosnia and Herzegovina, Georgia, Kosovo, North Macedonia, Moldova, Montenegro, Norway, Serbia, Switzerland, Turkey, Ukraine.
Orange: United Kingdom, separate cosmetics regulatory framework following Brexit
Since Brexit, the United Kingdom has established its own regulatory framework (UK Cosmetics Regulation). While it is still largely based on the former EU system, it is now a separate regime with its own notification portal (SCPN) and Responsible Person requirements.
More information on this matter here.
Before selling cosmetic products in the European Union, you must ensure that the product complies with Regulation (EC) No 1223/2009. The EU system is built around clear legal obligations that apply before the product is placed on the market.
Only cosmetic products for which a legal or natural person is designated within the EU as the Responsible Person may be placed on the EU market. In addition, the product must be supported by the required safety documentation and notified through the Cosmetic Products Notification Portal (CPNP).
Core EU Compliance Reviews
Before placing a cosmetic product on the EU market, companies typically need to complete the following core compliance reviews.
- Formula Review: Verification that the composition complies with the ingredient restrictions and prohibitions laid down in the Cosmetics Regulation.
- Laboratory Testing: Testing may be needed depending on the nature of the product and its risk profile, for example stability, compatibility, and microbiological quality.
- CPSR A & B (Cosmetic Product Safety Report): The product must be covered by a Cosmetic Product Safety Report prepared in accordance with Regulation (EC) No 1223/2009.
- Label and Claims Review: The label and product claims must comply with the applicable legal requirements before market placement.
EU Regulatory Documentation
The main EU documentation and notification elements include the following.
- PIF (Product Information File): the PIF must be available for the product and contain the information required by the Cosmetics Regulation, including the Cosmetic Product Safety Report and relevant product data.
- EU CPNP Notification: the product must be notified through the EU Cosmetic Products Notification Portal before it is placed on the market. Once notified in the CPNP, no additional national notification is required within the EU.
EU Responsible Person
The EU Responsible Person is a central legal requirement under Regulation (EC) No 1223/2009. Only cosmetic products for which a Responsible Person is designated within the EU may be placed on the market.
The Responsible Person is responsible for ensuring that the product complies with the applicable requirements of the Cosmetics Regulation, including product safety, PIF availability, notification, and cooperation with competent authorities.
Additional National Specificities
In the European Union, Regulation (EC) No 1223/2009 applies directly across all Member States. However, Member States may still have additional national rules in areas outside the core scope of the Cosmetics Regulation, for example environmental or waste related obligations.
US Cosmetics Regulation in March 2026: Less prescriptive than the EU, but no longer light touch
At federal level, U.S. cosmetics law still differs fundamentally from the EU model. The United States does not use the same pre market structure based on an EU style Responsible Person, PIF, and centralized pre market notification. However, that does not mean that U.S. compliance is simple or optional.
Since MoCRA, the U.S. cosmetics framework includes mandatory Facility Registration, Cosmetic Product Listing, serious Adverse Event Reporting, safety substantiation obligations, and broader FDA authorities in specific cases. Companies placing cosmetics on the U.S. market should therefore assess federal compliance much more carefully than under the pre MoCRA framework.
What MoCRA Changed in Practice
On December 29, 2022, the Modernization of Cosmetics Regulation Act of 2022 introduced the most significant expansion of FDA’s authority over cosmetics since 1938. In March 2026, MoCRA is no longer just a recent legislative development. Its operational framework is already active.
In practice, this means that many companies must now assess whether they have mandatory Facility Registration obligations, whether their products must be listed with FDA, whether they have adequate safety substantiation records, whether they are prepared to submit serious Adverse Event Reports within the statutory deadlines, and whether they can respond appropriately if FDA seeks access to records in the circumstances allowed by law.
Facility Registration and Product Listing
Under MoCRA, manufacturers and processors must register their cosmetic facilities with FDA and renew that registration every two years. In parallel, the responsible person must list each marketed cosmetic product with FDA, including product ingredients, and provide updates annually.
These obligations should not be confused with product approval. Cosmetic Product Facility Registration and Cosmetic Product Listing are not an FDA approval program, and FDA does not issue certificates for them.
The U.S. Responsible Person Under MoCRA
The U.S. responsible person is not the same concept as the EU Responsible Person. Under MoCRA, the responsible person means the manufacturer, packer, or distributor whose name appears on the cosmetic label.
This U.S. responsible person has specific statutory duties, including Cosmetic Product Listing and serious Adverse Event Reporting. However, the U.S. model does not create the same single market access gatekeeper role as the EU Responsible Person system.
From a European perspective, the U.S. federal framework may appear less prescriptive than the EU Cosmetics Regulation. However, companies should not confuse a less detailed statutory architecture with low compliance risk. U.S. compliance still requires careful assessment of product classification, labeling, ingredient permissibility, safety substantiation, Adverse Event processes, and MoCRA registration and listing obligations where applicable.
In practice, companies marketing cosmetics in the United States should be prepared to support product safety, substantiate claims where needed, maintain compliant labeling, and respond appropriately if FDA requests access to records under the authority provided by the FD&C Act as amended by MoCRA.
Core Federal U.S. Compliance Points
At federal level, companies should assess at least the following points before marketing cosmetic products in the United States:
- Product classification under U.S. law.
- Ingredient and color additive compliance.
- Labeling compliance under the FD&C Act and the Fair Packaging and Labeling Act.
- Adequate safety substantiation.
- Serious Adverse Event Reporting procedures.
- Facility Registration and product listing obligations where applicable.
- Record readiness if FDA requests access under the law.
From VCRP to Mandatory MoCRA Registration and Listing
The former Voluntary Cosmetic Registration Program is no longer the right reference point for describing current U.S. cosmetics compliance. FDA stopped accepting and processing VCRP submissions on March 27, 2023 as it moved to the MoCRA registration and listing framework.
Today, the relevant question is not whether a company wants to use the VCRP. The relevant question is whether the facility and the product fall within MoCRA’s mandatory registration and listing framework or within one of the statutory exemptions.
Federal and State Level Requirements
State level rules remain important in the United States, especially in areas such as ingredient restrictions, warnings, reporting, and environmental compliance. California remains particularly important in practice. However, since MoCRA, the federal baseline has become more significant than it was before.
Companies should therefore assess U.S. compliance on two levels: federal cosmetics requirements and any additional state specific obligations that may apply to the product.
| Key comparison points | EU | US |
|---|---|---|
| Regulated substances | Extensive positive, restricted, and prohibited substance framework | Much shorter express federal prohibited and restricted substance framework, but broader compliance analysis still required |
| Product information file | Yes | No direct EU equivalent |
| Notification, registration, and listing | CPNP notification before market placement | Facility registration and cosmetic product listing under MoCRA where applicable |
| Responsible person concept | Mandatory EU Responsible Person as a condition for placing the product on the market | Responsible person under MoCRA is the label named manufacturer, packer, or distributor, but the concept is not equivalent to the EU Responsible Person |
| Animal testing | Marketing ban linked to the EU framework | No equivalent general federal cosmetics ban in the same form |
| Nanomaterials | Specific notification and labeling rules apply | No equivalent standalone cosmetics nanomaterial notification regime comparable to the EU system |
| Good manufacturing practices | GMP based compliance is an established part of the framework | MoCRA requires FDA GMP regulations, but the system should not be presented as equivalent to the EU framework in the same way |
| Marketing claims | Specific claims framework, including Regulation (EU) No 655/2013 | Claims must not be false or misleading and must not turn the product into a drug |
Labeling Rules: EU vs U.S.
Labeling standards differ significantly between the EU and the United States;
Business information: the EU label must reflect the Responsible Person and the required mandatory particulars under the Cosmetics Regulation. In the U.S., labeling follows the FD&C Act and the Fair Packaging and Labeling Act framework, with a different structure.
Warnings: both systems may require warnings, but the EU framework is generally more specific and standardized for cosmetic labeling.
Net contents: the EU uses metric expression, while U.S. labeling follows U.S. customary presentation rules, often alongside metric information in practice.
Ingredient listing: in the EU, ingredient labeling follows the Cosmetics Regulation and includes specific rules for matters such as nanomaterials and fragrance allergens. The fragrance allergen regime was strengthened by Commission Regulation (EU) 2023/1545. In the U.S., ingredient labeling is governed by a different and generally less granular structure.
Claims and Product Classification
Both the EU and the United States prohibit cosmetic claims that are misleading or that push the product outside the legal definition of a cosmetic product. However, they do so through different regulatory structures.
In the EU, claims are assessed in a more explicit legal framework, including Regulation (EU) No 655/2013. In the United States, claims must not be false or misleading and must not cause the product to be regulated as a drug. Separately, MoCRA introduced a requirement for adequate safety substantiation, which should not be confused with general marketing claim substantiation.
Recommendations for Global Compliance
For brands selling in both the EU and the United States, the safest strategy is usually to assess each market separately rather than assuming that compliance in one jurisdiction automatically ensures compliance in the other.
In practice, many companies choose to align products with the more structured EU framework where possible, while still carrying out a separate U.S. review focused on product classification, labeling, safety substantiation, registration and listing obligations, and any relevant state level rules.
How Biorius Can Help
Understanding and applying cosmetics regulation across multiple markets requires more than a basic legal comparison. Biorius supports brands with EU and U.S. cosmetic compliance through services such as formula review, labeling and claims review, safety documentation support, Responsible Person services in the EU, and support related to U.S. federal and state level cosmetics requirements.
Author

Christophe Brault-Chevalier
Related Posts
11 March 2026
Get in Touch
Need a piece of advice, a quotation or answers to your questions?
Fill in this form or contact us directly: info@biorius.com – We will answer as soon as possible!
As specialists in cosmetic Regulations for more than 15 years, Biorius offers a reliable turnkey solution for placing cosmeticproducts in various markets:
-
First Class specialists in cosmetic regulations in Europe, the United Kingdom, the United States, and in more than 60 countries -
50 regulatory experts, toxicologists, pharmacists, and chemists to serve you. -
A unique model that guarantees you both the fastest turnaround possible and high-quality services. Curious? Ask us to know more! -
A best-in-class IT tool, free of charge, and saving a lot of your time. -
More than 1,500 international clients have already chosen Biorius! -
We evaluated more than 100,000 products and have always successfully managed compliance over 15 years of existence
Get in Touch
"*" indicates required fields
