Following a SCCS (the Scientific Committee on Consumer Safety) conclusion, the Commission Regulation (EU 2022/1181) decreases the Formaldehyde threshold for the labelling of a warning and changes the warning from ‘contains formaldehyde’ to ‘releases formaldehyde’.
Indeed, the SCCS considers that the present threshold (0.05%) does not sufficiently protect consumers sensitised to formaldehyde from exposure to free formaldehyde from formaldehyde releasers. However, their report highlights that reducing this threshold by a factor of 50 (that is 0.001% (10 ppm)), will protect the vast majority of consumers sensitised to formaldehyde.
This new threshold applies to the total free formaldehyde irrespective of whether a product contains one or more formaldehyde releaser(s).

The formaldehyde-releasing preservatives listed in the Annex V of the Regulation (EC) No 1223/2009 are as follows:
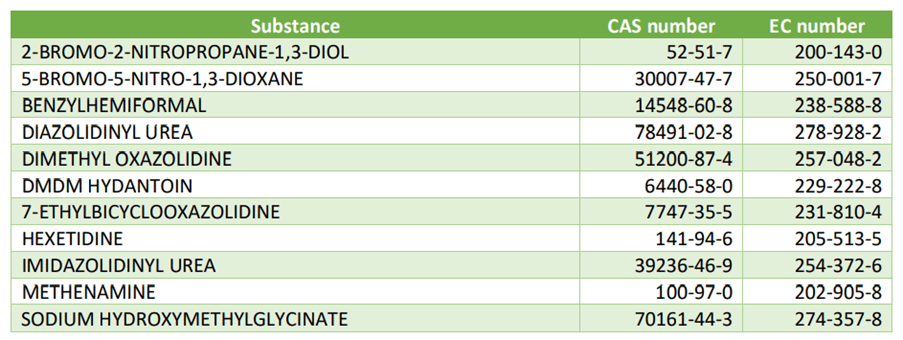
Following these conclusions, the European Commission (EC) stated that all finished products containing substances which are listed in Annex V of the Cosmetic Regulation and which release formaldehyde shall be labelled with the warning “releases formaldehyde” where the total concentration of formaldehyde released in the finished product exceeds 0,001 % (10 ppm).

The application deadlines are as below:
- From the 31st of July 2024 only products complying with the new restriction can be placed on the Union Market.
- From the 31st of July 2026 only products complying with the new restrictions can be further made available on the Union Market.
Contact us
For more information, do not hesitate to contact CRM (Customer Relationship Manager), if you are already a Biorius’ client or to contact us at info@biorius.com.


