
According to the Directive (EU) 2019/904 on the reduction of the impact of certain plastic products on the environment and its related Regulation (EU) 2020/2151, some considerations must be made regarding their future impact on cosmetics products.
The goal of this Directive – also known as Single-Use Plastic (SUP) Directive – is to reduce plastic waste and its devasting effects on the environment and human safety, while promoting a shift towards an innovative, sustainable and circular economy.
The biggest impact this Directive and its implementing Regulation will have in regards to cosmetics products is on the labelling requirements of wet wipes, i.e., pre-wetted personal care items.
Any personal care product that is designed to be used once, that contains plastic and that is pre-wetted falls under the scope of this Directive. Clarification on concerned categories will be made through various examples by the upcoming Commission Guidelines on the SUP Directive. The below marking must be displayed on the packaging of single-use pre-wetted wipes containing plastic:
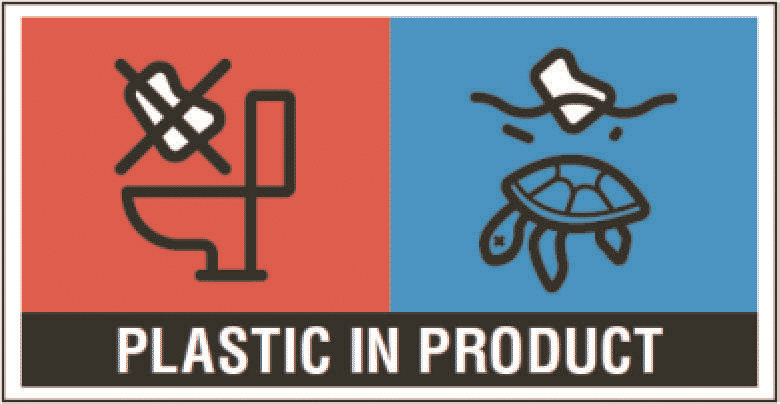
The information text of the marking shall be written in the official language or languages of the Member State(s) where the single-use plastic product is placed on the market.
The Commission Implementing Regulation (EU) 2020/2151, gives details on harmonized marking specifications – like position, size and design.
This marking shall be mandatory from 3 July 2021. However, a derogation is foreseen: the marking of packaging of wet wipes placed on the market before 4 July 2022 may be affixed by means of stickers.
For more information on this topic and all specifications like position, size and colors of this marking, check the article The Impact of Directive EU No 2019/904 and Regulation EU No 2020/2151 on Cosmetic Products available on our website.
Biorius remains at your disposal for any questions related to this topic and we will ensure that you are informed about the latest updates and all regulatory developments related to Directive (EU) 2019/904 and Regulation (EU) 2020/2151.



